 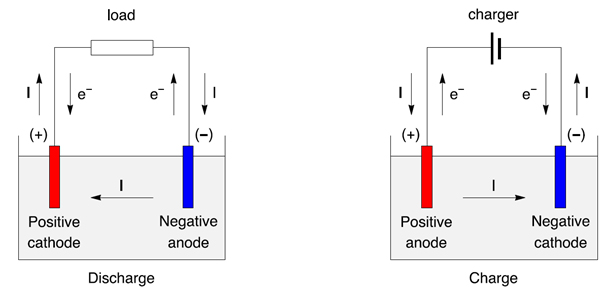 
At the same time, SO 4 reacts with copper anode and becomes CuSO 4 but in water it can not exist as single molecules instead of that CuSO 4 will split into Cu + +, SO 4 − − and dissolve in water. In the above process, after taking electrons the neutral copper atoms get deposited on the cathode. Electrolysis of Copper Sulfateīut since SO 4 radical can not exist in the electrical neutral state, it will attack copper anode and will form copper sulfate. This is the most basic principle of electrolysis. That means, current continues to circulate around the closed loop created by battery, electrolytic and electrodes. For continually taking and giving electrons in cathode and anode respectively, there must be flow of electrons in the external circuit of the electrolytic. In cathode, the positive cations take electrons from negative cathode and in anode, negative anions give electrons to the positive anode. The freely moving positively charged cations are attracted by cathode and negatively charged anions are attracted by anode. The electrode connected with negative terminal of the battery is known as cathode and the electrode connected with positive terminal of the battery is known as anode. These partly immersed rods are technically referred as electrodes. Now we will immerse two metal rods in the solution and we will apply an electrical potential difference between the rods externally by a battery. So, when any ionic bonded chemical is dissolved in water, the bonding strength between ions becomes much weaker and hence its molecules split into cations and anions moving freely in the solution. 
The relative permittivity of water is 80 at 20 oC. In ionic bond, the force acting between the ions is Coulombic force which is inversely proportional to the permittivity of the medium. Due to opposite charge both attract each other and form a bonding between them called the ionic bond. 
As a result, one atom becomes positively charged ion and another atom becomes a negative ion. In ionic bonds, one atom loses its valence electrons and another atom gains electrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
- Blog
- Home
- Star Projector Planetarium
- Last Battleground Survival Apk
- Pinball FX3
- Resident Evil 5 Youtube
- Steel Beasts Videos
- Mystik Belle Ending
- Child Of Light Review
- Her Story Boutique
- Tales Of Monkey Island Chapter 5
- Rodina Ahmed
- Tower Of Heaven Flash Game
- Bitlife Game
- Age Of War 2
- The Surge 2 Lifter Hook
- Crash Team Racing Download
- Mission Against Terror
- Tasty Town Game Olives And Chicken Recipe
- Aerotek
- I Am Innocent Michael Smith
- Star Projector Planetarium
- Mugen Rao
- The Crew 2 Car List
- Jet Set Radio
- Space Pirates And Zombies 2 Cheats
- Toontown Infinite
- Crafting Dead Mod
- Paradise Cove
- Pikmin 3 Enemies
- Death Mark Walkthrough
 RSS Feed
RSS Feed
